Trends in Epilepsy Research
Alana's story: Our journey to TSC care
11/13/2018
I will never forget the day I heard the words, “tuberous sclerosis complex.” I felt like it was the end of the world, but it was really just the beginning of a new kind of adventure for our family.
We were three months into trying to figure out what was “wrong” with our precious baby girl, Alana. She started having seizures in January 2014, and they were undoubtedly from an unknown cause.
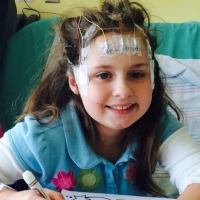
Lauryn's story: Living a seizure-free life after epilepsy surgery
11/13/2018
Oct 5, 2018.
If I could characterize our daughter’s journey to freedom from seizures, I’d call it a roller coaster of emotions. Thankfully, we felt safe and secure throughout the entire ride, all thanks to our phenomenal care team at Texas Children’s Hospital.
FDA approves epidiolex for epilepsy
06/26/2018
The Food and Drug Administration (FDA) has approved epidiolex, made of cannabidiol (CBD) and derived from marijuana plant to treat Dravet Syndrome and Lennox-Gestaut Syndrome, rare childhood-onset forms of epilepsy. Epidiolex, marketed by GW Pharmaceuticals Plc is the first cannabis-based drug to receive FDA approval. This decision could encourage more research into the medicinal properties of cannabis to treat other epileptic conditions.